Gas Dynamics &
|
Compressible FlowFluids are classified as either Incompressible or Compressible. Incompressible fluids do not undergo significant changes in density as they flow. In general, liquids are incompressible; water being an excellent example. In contrast, compressible fluids do undergo measurable density changes. Gases are generally compressible; air being the most well known compressible fluid. Compressibility of gases leads to many interesting features such as steady state shock waves, which are absent for incompressible fluids. In this section a broad introduction is given to the basics of compressible fluid flow.  Figure 1 : Classification of Fluids Though gases are compressible, the density changes they undergo at low speeds may not be considerable. Take air for instance. Fig. 2 shows the density changes plotted as a function of Mach Number. Density change is represented as $ρ\/ρ_0$ where $ρ_0$ is the air density at zero speed (i.e., Zero Mach Number). 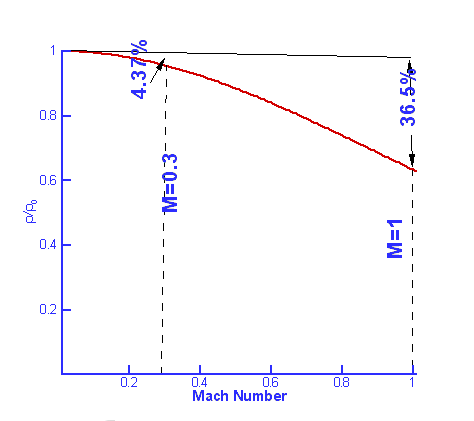 Figure 2 : Density change as a function of Mach Number For Mach numbers up to 0.3, density changes are within about 5% of $ρ_0$. So for all practical purposes one can ignore density changes in this region and analyse the flow as incompressible. But as the Mach Number increases beyond 0.3, changes do become appreciable and at a Mach Number of 1, the density change has reached 36.5%. At a Mach Number of 2, the density changes are as high as 77%. Another important difference between incompressible and compressible flows is due to temperature changes. For an incompressible flow temperature is generally constant. But in a compressible flow significant changes in temperature may occur leading to an exchange between the modes of energy. For an air flow at a Mach Number of 2 there are two important modes of energy; kinetic and internal. At this Mach Number, these can reach magnitudes of around $10^5$ Joules. When the Mach 2 flow is brought to rest, at a stagnation point, all the kinetic energy (motion) gets converted into internal energy (temperature). Consequently the temperature increases at the stagnation point. When a flow of Mach number 2 is at a temperature of $20^OC$ and then brought to rest, the stagnation temperature is as high as $260^OC$ as indicated in Fig. 3. 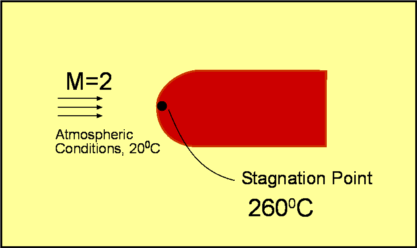 Figure 3 : Stagnation Temperature. A direct consequence of these facts is that when calculating compressible flows, the energy equation has to be considered (this was not done for incompressible flows). Further, to handle the exchange in modes of energy then the thermodynamics of the flow must be understood. Thermodynamics is a vast subject covering many topics. In this section just the basic concepts which are applicable to gasdynamics will be reviewed. System, Surroundings and Control VolumeConcepts in Thermodynamics are developed with the help of systems and control volumes. A system is an entity of fixed mass. Its boundary is not fixed and is allowed to vary depending upon the changes taking place within it. Considering the system shown below, namely water in a container placed on a heater, the choice of the system is made so as to obtain a simplified solution. The system could be defined as either (a - just the water) or (b - water plus container) or (c - water, container and surronding air) as in Fig. 4. Everything outside of the system becomes the surroundings. Properties of the system are usually measured by noting the changes it makes in the surrounding. For example, temperature of the water in system (a) is measured by the rise of the mercury column in a thermometer which is not part of the system. Sometimes the system and the surroundings are together called the universe. 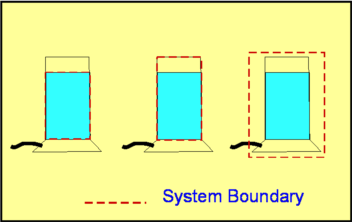 Figure 4 : Definition of a System Density, pressure, temperature, etc, become properties of a given system. Note that these are all measurable quantities. In addition, these properties also characterise a system. To define the state of a system (Fig. 5) uniquely we need to specify two properties say $P,T$ or $P,ρ$ or $T,s$ etc., where $P, T, ρ, s$ are pressure, temperature, density and specific entropy respectively.  Figure 5: State of a System Properties can be Extensive or Intensive. Extensive properties depend on the mass of the system. On the other hand, intensive properties are independent of the mass. Volume ,$V$, Energy, $E$, Entropy, $S$, Enthalpy, $H$ are Extensive properties. Corresponding intensive properties are Specific Volume, $v$, Specific Energy, $e$, Specific Entropy, $s$, and Specific Enthalpy, $h$, and are obtained by considering extensive properties per unit mass. In other words, $$v=V/m\text" , "e=E/m\text" , "s=S/m\text" , "h=H/m$$ Laws of ThermodynamicsThermodynamics centers around the following laws. Zeroth Law of ThermodynamicsThis laws helps define Temperature. It states - "Two systems which are in thermal equilibrium with a third system are themselves in thermal equilibrium." 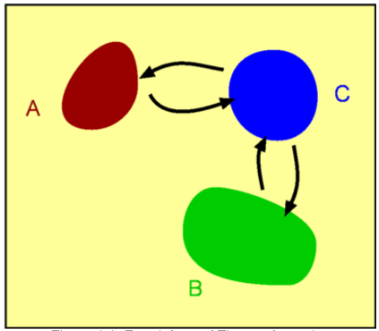 Figure 6 : Zeroth Law of Thermodynamics When in thermal equilibrium, it means that the two systems are at the same temperature. In the Fig 6, system A and B are independently in equilibrium with system C. It follows that A and B are themselves in thermal equilibrium and they are at the same temperature. First Law of ThermodynamicsThe first law of Thermodynamics is a statement of the principle of conservation of energy. It is simply stated as "Energy of a system and surroundings is conserved." Consider a system S. If an amount of heat per unit mass $dq$ is added into the system and the work done by the system is $dw$ per unit mass, then there is a change in internal energy of the system, $du$ given by, $$du = dq - dw$$ where $u$ is Internal Energy per unit mass or Specific Internal Energy. Using the following definition for Specific Enthalpy gives $$h=u+Pv = u+P/ρ$$ the statement for the first law can also be written as $$dh = dq + v.dp$$ In the above equation only one form of energy, internal, has been included. Other forms such as the kinetic energy have been ignored. Of course, it is possible to extend the analysis to account for all the forms of energy. Second Law of ThermodynamicsThe first law is a statement that energy is conserved during a process. It does not "worry" about the direction of the process whereas the Second Law does. The Second Law of Thermodynamcs determines the direction of a process. This requires an additional property, Entropy.  Figure 7 : Second Law of Thermodynamics There are many ways to state the second law. In this section, the version used is relevant to the study of gasdyanmics. Consider a Reversible Process. Suppose a system at state A undergoes changes, eg. by an addition of heat $Q$, and attains state B. While doing so the surroundings change from A' to B'. Let us try to bring the state of the system back to A by removing an amount of heat equal to $Q$. In doing so if we can bring the surroundings also back to state A' then the process is said to be reversible. This is possible only under ideal conditions. In any real process there is friction or other losses which dissipate energy. In reality, it is not possible to exactly bring the system back to state A and at the same time, surroundings back to A'. Assuming the process to be reversible, the second law defines entropy such that $$s_B-s_A=∫_A^B 1/T .dq$$ where $s$ is Specific Entropy. For small changes, the above equation is written as $$T.ds = dq$$ Generalising the equation, we have $$s_B-s_A≥∫_A^B 1/T .dq$$ where an '=' sign is used for reversible processes and '>' is used for irreversibe processes. Thus with any natural process, entropy of the system and universe increases. In the event the process is reversible entropy remains constant. Such a process is called an Isentropic process. Perfect Gas LawA perfect gas obeys the following law, provided it is only subject to isentropic processes, $$P=ρRT$$ where $R$ is the Gas constant. For a given gas, $R$ is given by $$R=Ʀ/M$$ where $Ʀ$ is known as the Universal Gas Constant and has the same value for all gases. Its numerical value is $8313.5 J\/kg-mol K$. $M$ is the molecular weight of the gas. The following table gives the value of the gas constant (along with other important constants) for some of the gases.
Consequences of First Law for a Perfect GasFor a perfect gas internal energy and enthalpy are functions of temperature alone. Hence, $$u = u(T)\text" , "h = h(T)$$ Specific Heat of a gas depends upon how heat is added - at constant pressure or at constant volume. We have two specific heats, $c_p$, specific heat at constant pressure and $c_v$, specific heat at constant volume. It can be shown that, $$c_p=({∂h}/{∂T})_P\text" , "c_v=({∂u}/{∂T})_V$$ The ratio of specific heats, $γ$, is in fact a measure of the number of degrees of freedom that the gas molecule possesses. Substituting gives,
$$R=c_p-c_v$$ A Calorically Perfect Gas is one for which $c_p$ and $c_v$ are constants. Accordingly, $$u=u(T)=c_vT\text" , "h=h(T)=c_pT$$ Consequences of Second Law for a Perfect GasFrom the First Law of Thermodynamics $$dh=dq+vdp$$ Now assuming a perfect gas and hence reversible processes, gives
$$dq=c_pdT-RT{dP}/P$$ Integrating between states 1 and 2, gives, $$s_2-s_1=c_p\ln(T_2/T_1)-R\ln(P_2/P_1)$$ If we assume that the process is isentropic or adiabatic (implying no heat transfer) and hence reversible, then the above equation leads to $$(T_2/T_1)=(P_2/P_1)^{{γ-1}/γ}=(ρ_2/ρ_1)^{γ-1}$$ A familiar form of equation for an isentropic flow is$$P/ρ^γ = \text"constant"$$ |